Hurricane Sandy's Rainfall Decoded Via CrowdSourcing
The storm smash-up that morphed Hurricane Sandy into a hybrid weather monster left a chemical trail that scientists have decoded with the help of crowdsourced water samples.
After a long run up the East Coast from its Caribbean birthplace, Superstorm Sandy was no longer considered a hurricane when it came ashore in New Jersey on Oct. 29, 2012. But the storm strengthened as it made landfall after colliding with a cold front sweeping in from the west.
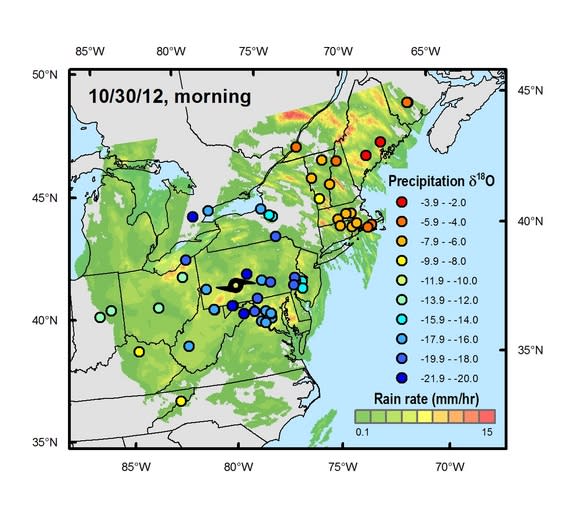
From oxygen and hydrogen isotopes (atoms of the same element with different numbers of neutrons) in rainfall collected by volunteers, University of Utah researchers tracked Hurricane Sandy's deadly march across the mid-Atlantic region. The rainfall isotopes reveal how the "Frankenstorm" sucked up moisture from both the incoming storm and the Atlantic Ocean, giving it new power to lash the Northeast with intense rainfall. The findings were published today (March 11) in the journal PLOS ONE.
"We have a really clear picture of what this storm looks like because we were able to involve so many people," said Stephen Good, lead study author and an isotope hydrologist at the University of Utah.
Crowdsourcing rain
A total of 685 rainfall samples were collected from more than 125 volunteers at sites from North Carolina to Indiana to New Brunswick, Canada. University of Utah professor Gabriel Bowen, a study co-author, led the call for samples on email lists, Twitter, Facebook, blogs and crowdsourcing sites as Hurricane Sandy approached the United States in late October.
"The idea was to try and get a full picture of what was happening isotopically in the storm," Good told Live Science. "We knew it was going to be a really important storm for a variety of reasons." [Infographic: Hurricane Sandy's Impact]
The new study offers one of the most detailed looks to date at a hurricane's changing isotopic patterns as these giant storms travel over land, Good said. Rainfall from hurricanes is noted for its exceptionally low levels of the isotope oxygen-18 — which is what the researchers found in water samples from the southern United States, where Sandy first hit the coast. When a cloud rains, the heavier isotopes fall first. Hurricanes drop so much rain that by the time they hit land, they're raining out much more of the lighter oxygen-16 isotope than the heavier oxygen-18 isotope. These storms also cannibalize their own precipitation, sucking up water with lighter oxygen isotopes.
Sandy's signature
But as Sandy traveled north and crashed into the cold front, its oxygen-18 levels rose. That's because the storm picked up new moisture, the researchers said. The rainfall samples also show a similar pattern for hydrogen isotopes, with a sudden uptick in deuterium (heavy hydrogen) over New England.
The shift in Hurricane Sandy's isotope patterns will also help the search for past hurricanes, the researchers said. For example, the low oxygen-18 isotope signature in hurricane rainfall leaves behind clues that geochemists can find in rocks and sediment. But Sandy shows this pattern can change if big storms hit other weather fronts.
"A really strong spatial gradient [in the isotopic record] can exist across a very large storm like this," Good said. "When we're trying to reconstruct information about previous large systems that have impacted the Northeast, a single reconstruction from a particular location might not tell the whole story."
Email Becky Oskin or follow her @beckyoskin. Follow us @livescience, Facebook & Google+. Original article on Live Science.
Copyright 2014 LiveScience, a TechMediaNetwork company. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.