Alaska health-care worker has severe allergic reaction to Pfizer COVID-19 vaccine
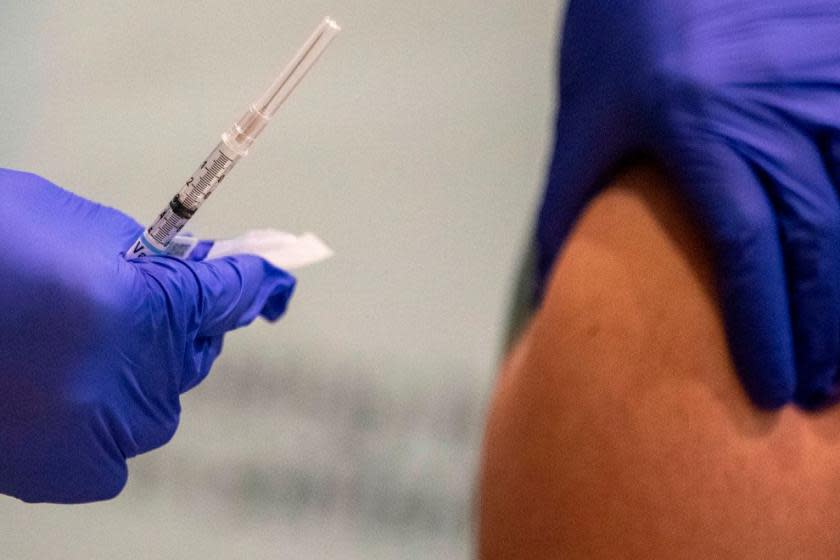
A health-care worker in Juneau, Alaska, had a severe allergic reaction to the Pfizer-BioNTech COVID-19 vaccine Tuesday, the first such reaction in the nascent U.S. coronavirus inoculation effort, Alaska officials said Wednesday. The unidentified woman, described as middle-aged with no history of allergies, had an anaphylactic reaction about 10 minutes after getting the shot at Juneau's Bartlett Regional Hospital. She started flushing, a rash appearing on her face and torso, said Lindy Jones, the attending physician who treated the woman.
"When she arrived at the emergency room she was feeling short of breath," Jones said. "She was not wheezy. Her heart rate was elevated." She responded to epinephrine, but then the symptoms reappeared, so she was monitored overnight in the intensive care unit. She has been discharged from the hospital.
A second health-care worker experienced more mild symptoms at the same hospital on Wednesday — puffy eyes, lightheadedness, and a scratchy throat — 10 minutes after the shot. He was treated with epinephrine, Pepcid, and Benadryl, and was back to normal within an hour, the hospital said. Those were the only two adverse reactions out of 144 doses administered at the hospital as of Wednesday night, and Alaska health officials said they have no plans to modify the vaccination campaign.
Federal health officials lauded Alaska for following guidelines advising that everyone inoculated with the new vaccine be monitored for 15 minutes, and anyone with a history of severe allergic reactions should stay under observation for 30 minutes after the injection. Pfizer did not report any allergic reactions in its large trial of the vaccine, but the company didn't allow people with histories of severe allergic reactions to participate in the trials. American doctors knew to look out for allergic reactions after two British health-care workers experienced them last week.
More stories from theweek.com
5 insanely funny cartoons about Trump's election-fraud failure
7 feel-good cartoons about the COVID vaccine breakthrough
Trump's ultimate self-own
