FDA advisors call for an end to never-ending booster shots as they try to map out a strategy for living with COVID
The FDA and CDC both say that the current COVID booster strategy is "stopgap" and not sustainable.
Instead, new vaccines are needed that target multiple variants, or stop infections at the nose.
An independent advisory committee to the FDA is weighing which vaccine strategies to try next, and how.
The nation's top disease and vaccine experts are trying to figure out their next move against COVID-19, before it's too late.
On Wednesday, an independent advisory committee to the US Food and Drug Administration — the agency entrusted with ensuring the country's COVID-19 vaccine supply is not only safe, but also useful and well-updated — met to discuss the future of COVID-19 vaccines and boosters. It was a departure from the committee's previous meetings during the pandemic, which have focused more deliberately on OK'ing specific COVID-19 vaccine candidates for different age groups, as well as booster shots.
The committee discussed:
When the US will know it's time for a brand new COVID vaccine that targets more recent variants, or gives people broader, stronger immunity, and how best to move quickly if that happens
How the government (instead of pharmaceutical companies) can drive the agenda on future vaccines, and what the vaccines should look like (nasal? multi-strain? universal?)
Whether there should be an annual COVID booster campaign each fall.
"There is a worry that protection against severe illness won't hold up forever," the FDA's Jerry Weir, who directs the Division of Viral Products Office of Vaccines Research and Review, said at the meeting.
The FDA and its advisors are already under a time crunch, to prepare for the possibility that new kinds of shots could be needed as early as this fall. Dr. Peter Marks, who directs the Center for Biologics Evaluation and Research (the arm of the FDA that is in charge of regulating all vaccines) acknowledged the current COVID vaccine strategy of boost-every-few-months with the original vaccine recipe — based on virus sequenced from Wuhan, China in early 2020— is not sustainable.
"We simply can't be boosting people as frequently as we are," he said at the meeting, calling the FDA's decision in late March to authorize second booster doses for people over 50 years old in the US a "stopgap measure" to protect highly vulnerable populations from severe disease, with the Omicron variant still spreading.
The argument for new vaccines, instead of boosting with what we have

Already, successful COVID-19 vaccine makers including Moderna and Pfizer are charting their next moves against the virus, without any direction from the federal government.
They are formulating new vaccines that would target the Omicron variant, or, potentially, future variants that might come our way. Other vaccine makers, including some which haven't created any successful COVID-19 vaccines yet, are also trying out different approaches to fighting this disease, in both early- and late-stage clinical trials.
A more cohesive national strategy for new vaccines is needed, the FDA committee members said. Tailoring vaccines too tightly to circulating variants, which change over the course of weeks and months, is a futile strategy that will lead vaccine makers to "miss the boat," committee member Dr. Michael Nelson, chief of the asthma, allergy, and immunology division at UVA Health, said.
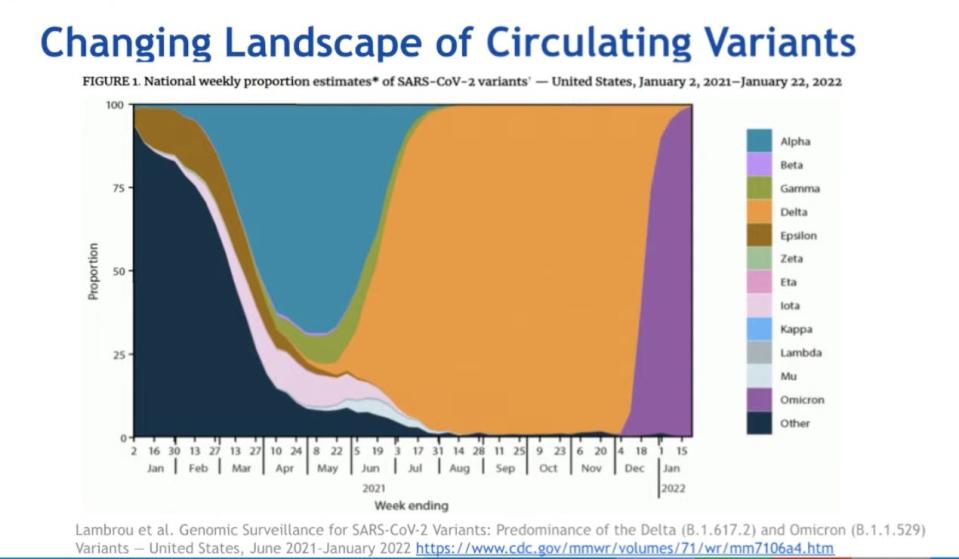
Nelson suggested, instead, "making changes only when we feel confident it's going to substantially lead to a longer duration" of protection, by "providing broad immunity against multiple variants."
Some vaccine makers, like Professor Drew Weissman at the University of Pennsylvania, are already working on that task. But large-scale human trials of those pan-corona vaccines are likely months, if not years away.
"We have to do tremendous work in researching more advanced vaccines, mucosal vaccines, pan-coronavirus vaccines," Marks said. "But we're not gonna get there for this coming year, so this is really trying to do the best we can with the knowledge we have."
Some experts say we may not need new shots
The COVID-19 vaccines authorized and approved for use in the US are still around 90% protective against severe infections leading to hospitalization or death, across all age groups.
"What is enough?" committee member Dr. Amanda Cohn, the chief medical officer at the National Center for Immunizations and Respiratory Diseases of the Centers for Disease Control and Prevention said at the meeting.
The CDC presented data at the meeting showing no evidence of waning vaccine effectiveness against hospitalization during the recent Omicron wave. Vaccine effectiveness against hospitalization in the real world remains well above 80%, even in highly vulnerable populations, including older adults and those with comorbidities.
"I think we may have to accept that level of protection, and then use other alternative ways to protect individuals with therapeutics and other measures," Cohn said. "I do not believe that boosters every 8 weeks, or even 4 months, is a long term strategy for prevention."
Either way, we need a better way to measure immunity, experts say
If we are going to go down the route of creating a new vaccine, experts on the committee urged the FDA to determine better "correlates of protection" for COVID-19 — i.e. finding a reliable way to measure COVID-19 immunity levels in people, to determine when they're decently protected against the virus.
Measuring antibody levels, as is done so routinely now, is a crude measure of protection, a "poor man's" indicator of immunity against COVID, Marks said during the meeting.
The problem with measuring antibodies is that experts know it's imprecise, which means there's no surefire way to know if someone really needs a booster or a new shot, or what kind of protection they can expect to derive from it.
Dr. Ofer Levy, director of the precision vaccines program division of infectious diseases at Boston Children's Hospital, suggested the US government should develop a repository, where the very best, most high-quality immunology studies from around the world could be consulted. That, he said, could help drive the conversation about what the next vaccines need to do for our immune systems, and how we'll know when they're working well.
Advisors will meet again in summer to decide a plan for the fall
The FDA advisory committee is expected to meet again some time this summer, hopefully with more data from pharmaceutical companies about next-generation vaccine candidates they're working on. Then, the hope is that the committee can make some informed decisions about what should happen this fall and beyond, assessing whether additional shots will be needed, and if so when, for whom, and what kind.
"We're in uncharted territory," committee chair Dr. Arnold Monto said, expressing the difficulty of the task. "With SARS-CoV-2, a lot of things have happened that have never happened before."
Read the original article on Business Insider

