FDA Authorizes Moderna and Pfizer Vaccines for Kids as Young as 6 Months

Updated, June 17
The Federal Food and Drug Administration on Friday authorized both Moderna’s and Pfizer-BioNTech’s vaccines for children as young as 6 months old — meaning COVID shots for the last age group of Americans without access may be just days away.
The vaccines now await sign-off from the Centers for Disease Control and Prevention, the final step before children under 5 can begin rolling up their sleeves. The White House expects providers to begin administering doses on Tuesday, immediately after the federal Juneteenth holiday.
On Wednesday, an FDA panel of experts — made up of pediatricians, infectious disease experts and vaccine researchers — voted 21-0 in favor of both vaccine options.
“There are so many parents who are absolutely desperate to get this vaccine, and I think we owe it to them to give them the choice,” said committee member Jay Portnoy, a professor of pediatrics at Children’s Mercy Hospital in Kansas City, Missouri.
Philip Chan, medical director for the Rhode Island Department of Health, called the panel’s unanimous vote a “major milestone in COVID prevention.” His state is ready to distribute the shots as soon as they receive the all clear, he said.
“We’ve pre-ordered thousands of [doses], and we expect them to ship within 24 or 48 hours after the FDA issues the EUA” (emergency use authorization), he told The 74 Wednesday. With EUA now granted, the COVID shots should soon be on their way.
Parents who are eager to finally vaccinate their young children took advantage of the public comment period at the FDA’s Wednesday committee meeting to urge advisors to recommend authorization. Michael Baker, the father of a 1 and a 3 year old, described the tough choices he has had to make to protect his children from the virus. He shared a slide of all the events they have missed out on during the pandemic, including weddings, holidays and funerals.
“All I am asking is now that … I have the choice to vaccinate my children, [that] I have the choice to do it in the most timely fashion possible,” he said to the committee.

Parents like Baker awaiting vaccines for their little ones have been on a months-long rollercoaster that has repeatedly raised their hopes only to later send them crashing down. In late February, Pfizer-BioNTech first submitted a request asking the FDA to grant emergency authorization for a two-dose regimen of their vaccine for children 6 months to 4 years old, only to then withdraw the application just five days later. Then in April, when Moderna was on the verge of submitting its EUA application for the age group, the FDA postponed the committee review process until Pfizer’s shots were also ready.
Just 29% of children 5 to 11 years old and 59% of youth 12 to 17 years old have so far received two vaccine doses, according to data from the American Academy of Pediatrics.
However, Katelyn Jetelina, creator of Your Local Epidemiologist, wrote in her hyper-popular newsletter that parents of young kids should still take COVID seriously and vaccinate their children.
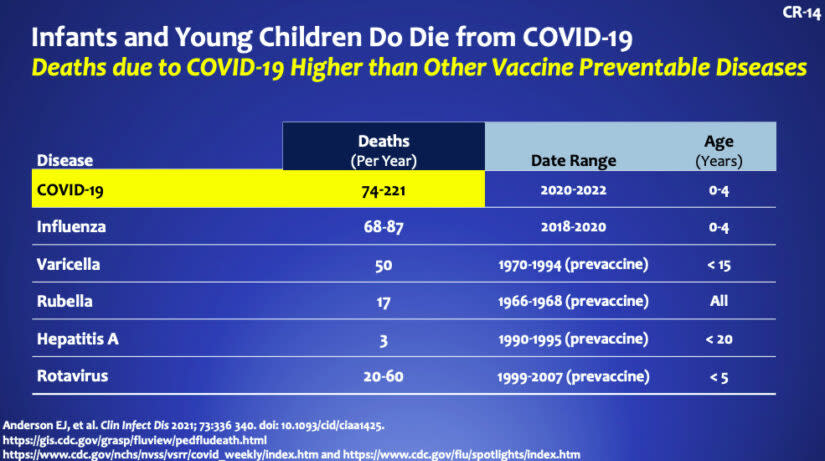
“The rate of severe disease is lower compared to adults, but this is an inherently flawed comparison because kids don’t die as often as adults. Since the beginning of the pandemic, 442 children aged 0-4 years old have died from COVID-19. If we compare to other vaccine preventable diseases among children, deaths due to COVID19 are highest. We cannot become numb to these deaths,” she explained.
COVID cases across the U.S. are finally leveling off after a springtime surge fueled by an Omicron subvariant spurred bumps in pediatric infections and hospitalizations through the final weeks of the school year.
You can view the FDA’s slides and watch the full recording of its June 15 vaccine advisory committee’s virtual meeting:

