Moderna just finished recruiting 30,000 people for its coronavirus vaccine trial. One graph reveals how the biotech slowed down its research to recruit more minorities.
The coronavirus vaccine frontrunner Moderna has finished enrolling volunteers for its final clinical trial, the biotech said Thursday.
The 30,000-person study is expected to produce data showing whether or not the vaccine prevents symptomatic COVID-19 cases before the end of this year.
Moderna CEO Stephane Bancel has previously told Business Insider he expects efficacy data to come in November.
The Massachusetts biotech also slowed enrollment in the final weeks of recruiting volunteers for this study to recruit more minorities.
Overall, 37% of the study's population is from minority communities, generally reflecting the diversity of the US, according to the company.
For more stories like this, sign up here for Business Insider's daily healthcare newsletter.
The Massachusetts biotech Moderna has finished recruiting volunteers for the final stage of clinical testing for its coronavirus vaccine candidate, the company said Thursday.
The end results are promising on a key challenge that faced vaccine-makers: recruiting a diverse sampling of Americans that includes representative numbers of minority groups. Vaccine researchers have feared that studies that are overwhelmingly white wouldn't be able to ultimately persuade minority groups that a shot works in their communities.
Moderna's study recruited 30,000 volunteers, who are randomized to receive either the two-dose regimen of the biotech's experimental shot or placebo injections of saltwater. Results from the trial will show if the vaccine can prevent symptomatic COVID-19 cases, and the company anticipates efficacy results to come in November.
About 11,000 of the participants are from minority communities, making up 37% of the trial. That includes more than 6,000 people who identify as Hispanic or Latinx and 3,000 who identify as Black or African American, Moderna said.
It was far from a foregone conclusion that this pivotal study would be diverse. Recruiting minority groups has been a longtime failure of the drug industry. The early coronavirus vaccine studies, for instance, were about 90% white and less than 1% Black among volunteers.
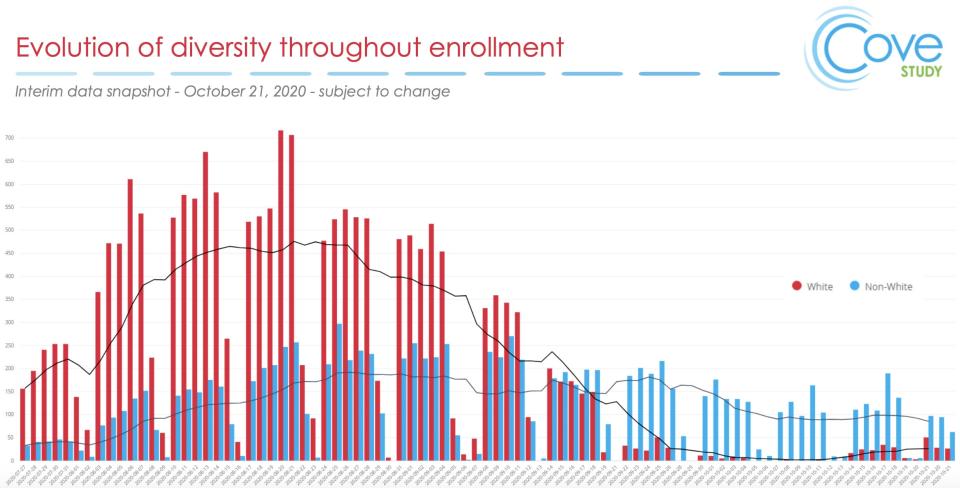
A new graph released Thursday shows Moderna's late-stage trial began recruiting mainly white people.
The chart shows a dramatic change in the final month of the trial, recruiting almost entirely non-white volunteers. The final trial population is 63% white, 20% Hispanic/Latinx, 10% Black or African American, and 4% Asian.
In mid-September, CEO Stephane Bancel told Business Insider the number of Black participants was "a bit too low," leading to the intentional slowdown. Moderna closed trial sites that had done a poor job enrolling people of color.
"We said let's slow down by basically shutting down the sites that have not done a good job of recruiting minorities, and let's favor the sites that have done a good job at recruiting minorities," he said.
"We want to make this vaccine so if it works and it is approved, people use it," Bancel added. "It would be really sad if one of the big communities of the country doesn't have enough representation, that people don't feel the vaccine is safe for their genetic pool of diversity, and effective."
Moderna is one of the two leading vaccine programs aiming for a potential emergency approval before the end of this year. The other leading COVID-19 vaccine program is led by Pfizer and BioNTech, which have enrolled just under 40,000 people in its study, according to an update given Monday. Pfizer's CEO has said the company may know if the vaccine works by the end of this month.
Read the original article on Business Insider

