Oxford-AstraZeneca Q&A: How effective is the Covid-19 vaccine, and how is it different to Pfizer?

The coronavirus vaccine rollout is in full flight in the UK.
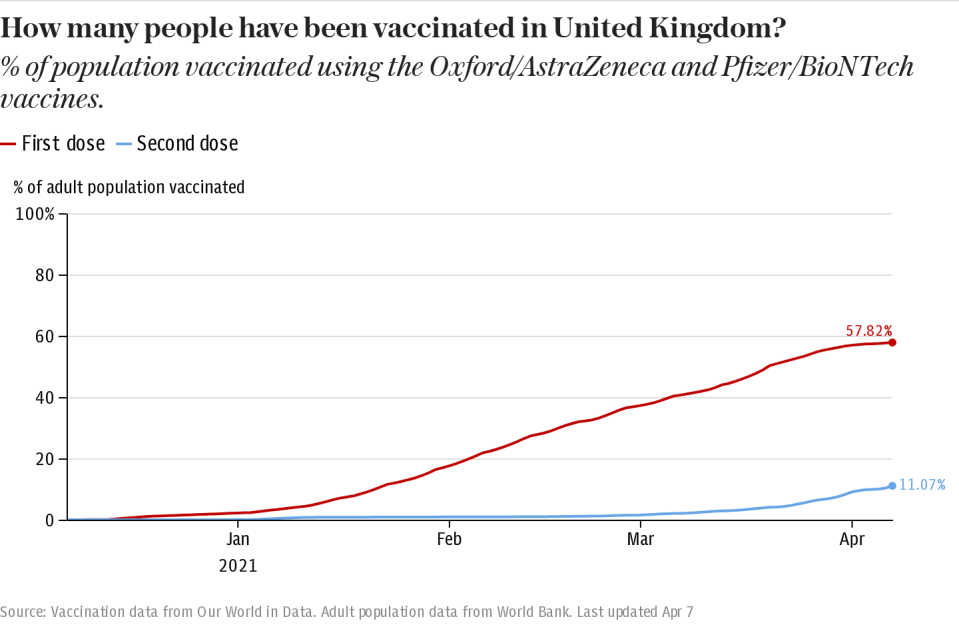
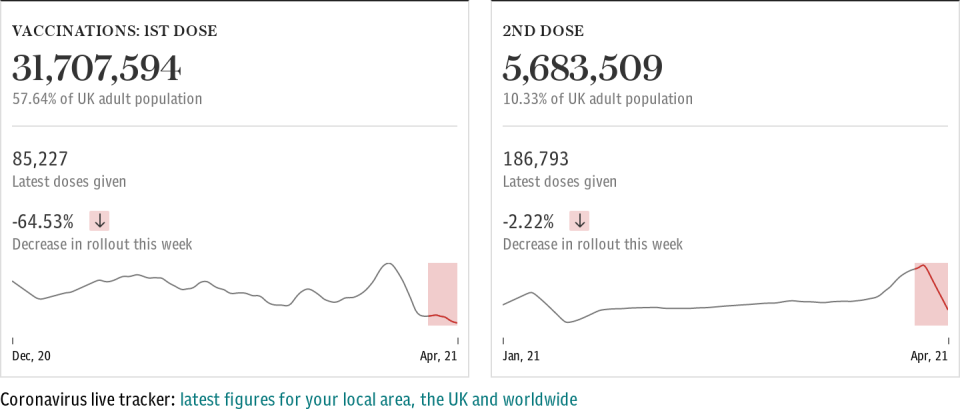
A total of 31,622,367 people have now received a first dose of the vaccine as of Apr 7, while 5,496,716 have had their second jab - this means around 59 per cent of all UK adults have now had at least one jab, while 10 per cent have had their second.
The number of first doses administered each day had been steadily climbing since December - reaching more than 400,000 a day mid-February.
Matt Hancock, the Health Secretary, said on Mar 28: I’m absolutely thrilled that more than 30 million people have now had the jab across the UK - including 650,000 vaccinations delivered yesterday.
"The vaccine is saving lives and is our route out of this pandemic. I want to say a massive thanks to the team. When you get the call - get the jab."
But after concerns about patients receiving the Oxford/AstraZeneca vaccine and a reported link to developing blood clots, the UK's medicines regulator hosted a joint briefing with the Government's vaccines advisory committee on Apr 7.
The briefing concluded that blood clots should be listed as a "very rare" side effect of the vaccine, and restated that the benefits continue to outweigh the risks.
In a rapid review of the jab European Medicines Agency declared it "safe and effective". In a briefing on April 7, the EMA concluded that blood clots should be listed as a "very rare" side effect, and restated that its benefits far outweigh any risk. Further to the review, the UK's Medicines and Healthcare Regulatory Agency stated that there is a four in one million chance of getting a blood clot from the AstraZeneca vaccine. Despite the negligible risk, those aged under 30 in the UK will be offered Pfizer or Moderna instead.
Oxford University had recently announced that it would pause vaccine trials in children until the Medicine and Healthcare products Regulatory Agency (MHRA) had taken a view on whether the jab was responsible for rare blood clots.
Adam Finn, a professor of paediatrics at the University of Bristol and who also sits on the Joint Committee on Vaccination and Immunisation (JCVI), said on Apr 7 that concerns over the Oxford jab are being taken "very seriously" and "very thoroughly" investigated.
However, he urged people being offered the vaccine at the moment to take it, saying the "risk-benefit is very strongly in favour of receiving the vaccine".
It comes as people in Wales have received the first doses of the Moderna vaccine, with the vaccines minister saying the jabs will be deployed widely "around the third week of April."
The Moderna vaccine has become the third in circulation in Britain, after the rollout of the Pfizer and AstraZeneca vaccines in December and January, respectively. It is expected the vaccine, which was found to be 94.1 per cent effective in phase three trials, will be rolled out in England in the next few days, as of Apr 7, according to small business minister Paul Scully.
Mr Hancock had previously said a "significant reduction" in jabs from Mar 29 meant the focus for the NHS in April would be on giving second doses to people who were vaccinated earlier in the year.
Sixty million doses of the Novavax jab have been secured by the UK, which Mr Zahawi was himself injected with as part of the trial, which was shown to be 89.3 per cent effective in preventing coronavirus in participants.
Covid lockdown roadmap: key dates revealed for easing restrictions in England
How does the AstraZeneca/Oxford vaccine work?
The vaccine – called ChAdOx1 nCoV-19 – uses a harmless, weakened version of a common virus that causes colds in chimpanzees.
Researchers have already used this technology to produce vaccines against a number of pathogens including flu, Zika and Middle East Respiratory Syndrome (Mers). The virus is genetically modified so it is impossible for it to grow in humans.
Scientists have transferred the genetic instructions for coronavirus's specific "spike protein" – which it needs to invade cells – to the vaccine.
When the vaccine enters cells inside the body, it uses this genetic code to produce the surface spike protein of the coronavirus. This induces an immune response, priming the immune system to attack coronavirus if it infects the body.
How effective is it?
Oxford University and AstraZeneca – a British-Swedish multinational pharmaceutical and biopharmaceutical company with its headquarters in Cambridge – announced that their jab was effective in preventing many people getting ill and it has been shown to work in different age groups, including the elderly.
Partial immunity against Sars-Cov-2 is then detected approximately 22 days after administration of the first dose, the MHRA reported.
Data published in The Lancet in early December showed the vaccine was 62 per cent effective in preventing Covid-19 among a group of 4,440 people given two standard doses of the vaccine, when compared with 4,455 people given a placebo drug. The overall Lancet data, which was peer-reviewed, set out full results from clinical trials of more than 20,000 people.
Among the people given the placebo drug, 10 were admitted to hospital with coronavirus, including two with severe Covid-19 which resulted in one death. However, among those receiving the vaccine, there were no hospital admissions or severe cases. The combined analysis from both dosing regimens resulted in an average efficacy of 70.4 per cent.
According to results, a single dose of the Oxford-AstraZeneca vaccine prevents two-thirds of Covid transmissions. The data, released on Feb 2, found a single dose was 76 per cent effective in fending off infection between 22 days and 90 days post-injection, rising to 82.4 per cent after a second dose at that stage.
In a major vindication of government policy, the study also found that the vaccine is more effective overall if the second dose is delayed from 28 days until 90 days.
This was further backed up by new evidence published on Mar 1. A new study from Public Health England (PHE) suggests that a single dose of the Pfizer or Oxford vaccine offers dramatic protection against hospital admission and severe disease in older people.
The PHE data, from more than seven million people, found that either jab cuts the risk of hospitalisation among over 80s by more than 80 per cent while reducing infections in such age groups by around two-thirds.
The AstraZeneca jab slightly outperformed Pfizer with the Health Secretary saying that the findings from 7.5 million people aged 70 and over showed the vaccination programme is saving lives, and may explain dramatically reduced pressures on hospitals.
The AstraZeneca vaccine is 100 per cent effective against severe or critical disease and hospitalisation, a US study published on Mar 22, has shown. Some 32,449 people across all age groups took part in the phase three trial in the US, Chile and Peru, with a total of 141 cases of symptomatic Covid-19 reported.
The study showed that the AstraZeneca/Oxford vaccine was 79 per cent effective at preventing symptomatic illness, and was 100 per cent effective against severe or critical disease and hospitalisation. The company later revised the 79 per cent figure down to 76 per cent after American authorities raised concerns that results reported from its US trial were outdated.
Is it effective against the new variants?
Many vaccine manufacturers have already announce plans to alter their jabs to take into account these variants, including the Oxford-AstraZeneca team. Prof Sarah Gilbert, who has headed up the development of this vaccine, has said a new version could be ready by autumn.
Some experts believe the Covid vaccine may have to be reformulated annually - like the flu vaccine - to combat what is known as “antigenic drift”, or the emergence of new variants.
A study carried out by the developers of the Pfizer-BioNTech jab found that it elicited good antibody responses against engineered viruses with similar mutations to the UK and South African variants. But other vaccines tested on populations where the variants were circulating - J&J, Moderna and Novavax - have shown they are less effective against the South African variant.
However, they all provide some protection and are more effective against severe illness.
South Africa announced that it was pausing its vaccination programme after a study found that the Oxford-AstraZeneca vaccine was not effective at preventing mild to moderate disease. However, many experts pointed out that this study was small and only looked at individuals aged under 65.
The Moderna and Pfizer vaccines are based on mRNA technology and are the easiest to tweak, with Dr Ugur Sahin, CEO of BioNtech, estimating it could take just six weeks to adapt the vaccine.
Most experts caution against viewing vaccines through an "all or nothing" prism – even if they offer only 50 per cent protection against the variants that will be better than not being vaccinated at all.
Second-generation vaccines are currently in development, with UK and German firms GSK and Curevac collaborating on a multi-valent vaccine that can be used against several strains.
Meanwhile Moderna and Pfizer-BioNTech both said their vaccines were effective against new variants of the coronavirus discovered in Britain and South Africa, but they are slightly less protective against the variant in South Africa, which may be more adept at dodging antibodies in the bloodstream.
As a precaution, Moderna has begun developing a new form of its vaccine that could be used as a booster shot against the variant in South Africa.
How is the rollout going?
The Oxford vaccine rollout began on Jan 4 at the Oxford University Hospitals NHS Foundation Trust's Churchill Hospital.
As of March 29, more than 30 million people in the UK have had at least one Covid vaccine since the programme was launched, while 3 million have received a second dose.
However, people under the age of 50 may now have to wait up to a month longer than planned for their Covid vaccination because of a major shortage of vaccines, NHS leaders have said.
The unexpected delay was revealed in a letter to health service chiefs, who have been ordered to stop booking first-dose appointments for anyone under 50 for all of April.
The letter from NHS England explained that the move was necessary because there would be a “significant reduction in weekly supply available from manufacturers beginning in the week commencing 29 March”.
The chief executive of Serum Institute of India (SII), now the world’s largest manufacturer of Covid vaccines, which has been running full tilt to produce 50m doses per month of the AstraZeneca/University of Oxford jab, told the Telegraph that the Indian government was to blame for the delay.
Officials are reported to be temporarily holding exports while the government decides on how to deal with a resurgence in Covid infections.
Additionally, the EU is currently discussing an export ban on vaccines to the UK, a decision which would risk 26 million Britons missing their second dose of the jab. The vaccine rollout of the UK has prioritised as many people receiving their first dose as possible, meaning there is currently no stockpile. This means that the current priority is ensuring people receive their second dose within the 12 week period.
Safety data on the critical child vaccine study being run by Oxford University – on which ministers are waiting before making their final decisions – is expected shortly, with its conclusions due in June or July.
A Department of Health and Social Care spokesman said "no decisions have been made on whether children should be offered vaccinations".
It comes after Prime Minister Boris Johnson received his first dose of the Oxford/AstraZeneca vaccine on Mar 19. Mr Johnson urged the public to keep faith with the Oxford/AstraZeneca vaccine, after regulators ruled the jab is safe and its benefits "far outweigh any risks."
Why is there concern over the AstraZeneca jab?
All the major European countries suspended the use of the AstraZeneca jab in early March after a flurry of nations announced temporary halts to their programmes.
Norway and Austria were the first to sound the alarm - Austria reported that a person was diagnosed with blood clots and died 10 days after they had received the vaccination. Another was hospitalised with pulmonary embolism (blockage in arteries in the lungs) after being vaccinated.
Then Denmark announced that someone had died after receiving the jab and became the first country to suspend its AstraZeneca programme.
This prompted other countries, including Norway, the Netherlands and Ireland to follow suit. Thailand also suspended its programme but has now restarted it.
However, many European countries said they would resume their Oxford/Astrazeneca vaccine roll out on Mar 18 after the European Medicines Agency (EMA) said its initial investigation of possible side effects has concluded the vaccine was “safe and effective” and was "not associated" with a higher risk of clots.
But on Apr 6, a senior official at the EMA said there is a link between the AstraZeneca vaccine and blood clots.
Marco Cavaleri, the head of vaccines at the EMA, told an Italian newspaper that an association was now "clear", but added that experts were still unsure what was causing the clotting.
Britain's Medicines Healthcare and Regulatory Agency (MHRA) is still investigating seven deaths and 30 cases of clotting in people who had recently had the AstraZeneca jab.
On Apr 5, Channel 4 News reported that the MHRA is considering banning the jab in younger people, with a decision expected this week. Prof Chris Whitty, the chief medical officer, last week said it may get to a point where the benefits of the jab no longer outweigh the risks for younger people.
Mr Macron prompted an outcry earlier this year when he claimed the jab was "quasi-ineffective" for pensioners.
In February, Mrs Merkel, 66, faced heavy criticism for refusing the AstraZeneca jab, insisting it was not recommended for people over 65.
On March 19, Kate Bingham, the former head of the Government's vaccine taskforce, accused the French president and German chancellor of encouraging anti-vaxxers after both cast doubt on the efficacy of the vaccine, developed by British scientists.
Bingham described the leadership as "completely irresponsible".
Just 12 per cent of the EU population has been vaccinated, and more than half the bloc's stockpile of around 15 million AstraZeneca jabs remains unused. Both France and Germany are experiencing third Covid waves, with Paris and other areas of France being locked down for a month.
Everything we know about the MHRA announcement and blood clots

Should we be worried?
Out of the 17 million people across Europe who have received the vaccine there have been 15 events of deep vein thrombosis and 22 events of pulmonary embolism.
Blood clots are a common condition, particularly among older people who are currently prioritised for vaccination.
Given that one in 1,000 people suffer from blood clots every year, we would expect that out of the 17 million who had been vaccinated 17,000 of those would get a blood clot over the next 12 months – about 320 people every week.
AstraZeneca has pointed out that in clinical trials there were fewer blood clots in those given the vaccine than in those who did not receive it.
Blood clotting is a very common condition in Covid patients and it is unclear whether any of the people reported to have died or suffered blood clots actually had the disease.
Given the tiny numbers of people who have suffered blood clots and the vast numbers who have had the vaccine the link seems very tenuous.
The MHRA have urged people to continue receiving the vaccine, emphasising that the chances of blood clotting are one in a million.
Read more: The link between the AstraZeneca vaccine and blood clots: why it is safe to have the jab
What have the experts said?
On March 18, the the European's Medicine Agency concluded that the AstraZeneca jab is a "safe and effective vaccine".
Emer Cooke, chief of the EMA, said the Oxford/AstraZeneca vaccine is not associated "with an increase in the overall risk of thromboembolic events and blood clots".
However, a senior official at the European Medicines Agency (EMA) said on Apr 6 that there is a link between the AstraZeneca vaccine and blood clots and that the rollout in younger people should be halted until further safety guidance was issued.
On Wed Apr 7, the European Medicines Agency concluded that blood clots should be listed as a "very rare" side effect of the vaccine, restating that the benefits continue to outweigh the risks.
The EMA stressed the importance of vaccination in the fight against Covid-19, stating that the AstraZeneca vaccine had been "proven to be highly effective".
Marco Cavaleri, the head of vaccines at the EMA, told an Italian newspaper that an association was now "clear", but added that experts were still unsure what was causing the clotting.
What has Germany said?
On Tuesday 30 March, Germany became the latest in a growing list of countries, including Canada and France, to suspend use of the vaccine in younger age groups. In Germany the cut off is 60, while in Canada and France only those age 55 and above are now receiving the jab.
Germany's medical regulator announced on Tuesday it had received a total of 31 reports of rare blood clots in recent recipients of the AstraZeneca vaccine. Nine died and all but two of the cases involved women aged 20 to 63, the Paul Ehrlich Institute said.
The issue involving blood clots associated with thrombocytopenia, a low blood platelet count, is the same one which caused many EU countries to temporarily halt the use of the jab earlier this month.
Based on the data available at the time, the European Medicines Agency (EMA) concluded there was not enough evidence to establish a link between the problem and the vaccine, and recommended it continue to be used. The benefits of inoculation greatly outweighed the risks for all age groups, it stressed.
On Tuesday, a team of European researchers published a not-yet-peer-reviewed preprint which looked at nine patients in Germany and Austria who developed thrombocytopenia and clots following vaccination.
“The AZD1222 vaccine is associated with development of a prothrombotic disorder that clinically resembles heparin-induced thrombocytopenia but which shows a different serological profile”, they said.
Not surprisingly, regulators in the UK, Europe and North America are now all racing to better understand if an association between the jab and the condition really exists and, if so, at what rate it occurs.
The relevant clotting data from Norway put the risk at about four in 100,000 and the German figures put it at just over one in 100,000.
The UK Government has insisted it is "100 per cent confident" in the effectiveness of the Oxford-AstraZeneca vaccine.
"We're 100 per cent confident in the efficacy of the vaccine, that's borne out by study after study, by our own independent world-class regulators and by recent research, for example, by Public Health England that's shown that thousands of people's lives have been saved since the start of this year alone thanks to our vaccine programme," Robert Jenrick, the communities secretary, said.
"People should continue to go forward, get the vaccine, I certainly will when my time comes, it is a safe vaccine and the UK's vaccine rollout is saving people's lives right across the country every day."
But for the world as a whole these reports could pose an even greater problem. Covax, the international facility distributing vaccine to the developing countries, is heavily reliant on AstraZeneca.
Related: Britain could be in for a rough ride with the Oxford-AstraZeneca jab, but there’s no need to panic
Why were there some concerns in the US?
On Tuesday 23 March, AstraZeneca was forced to defend data showing its Covid-19 vaccine was highly effective after US health officials suggested some results were based on "outdated information".
Hours after the UK-Swedish drug giant reported its jab provided strong protection among adults, the National Institute of Allergy and Infectious Diseases (NIAID) said it was worried the trial results have given only a partial picture.
The statement immediately threatened to overshadow results that had been viewed as good news for the company after public doubts over its vaccine, and mire the firm in fresh controversy.
Is the vaccine effective in those over the age of 65?
While some European countries were initially uncertain about the effectiveness of the jab in the over 65s, new data on the effectiveness of the AstraZeneca vaccine has "vindicated" the UK's decision to roll it out to older age groups, England's deputy chief medical officer has said.
Professor Jonathan Van-Tam said the UK's Joint Committee on Vaccination and Immunisation (JCVI) had taken the view that it was "not immunologically plausible" the vaccine would work in younger age groups and not older ones.
The UK remains confident that the vaccine is effective for over 65s, Munir Pirmohamed also told a MHRA news briefing.
"Since (initial approval) we've seen more data coming through from AstraZeneca as more people are completing the trial, which highlights again that efficacy in the elderly is seen, and there's no evidence of lack of efficacy," he said.
The Medicines and Healthcare products Regulatory Agency (MHRA) said its analysis showed a reporting rate of three suspected side-effects for every 1,000 doses of vaccine administered - with the issues reported being similar to those for the annual flu vaccine.
Dr June Raine, chief executive of the MHRA, said: "Vaccines are the most effective way to protect against Covid-19 and save lives and prevent serious complications from this terrible virus.
"The data we have collected provides further reassurance that the Covid-19 vaccines are safe and continue to meet the rigorous regulatory standards required for all vaccines.
"We remain confident that the benefits of these vaccines outweigh any risks."
Read more: German response to Oxford vaccine is unfair blow for UK scientists and will cause needless worry
Why was the EU arguing with the UK over vaccine exports?
The European Union and the UK were embroiled in a row over vaccine exports after the EU demanded access to AstraZeneca vaccines that were being manufactured in the UK.
This demand by the EU was an attempt to make up for a shortfall in their supply after the British-Swedish pharmaceutical company said it could only supply a quarter of the jabs - 31 million instead of the initial 80 million it had aimed for in the first quarter of the year.
While Britain used emergency procedures to grant market approval to the AstraZeneca vaccine, and signed a contract with the company three months before Brussels, the EU used a slower process overseen by the European Medicines Agency.

The contract between the EU and AstraZeneca that was agreed in August 2020 revealed that the two sides agreed manufacturing sites "shall include the United Kingdom" - adding weight to the EU's position. It also stipulates that other deals cannot supersede any agreement with the EU.
Brussels accused AstraZeneca of breaching its contract and, amid suspicions it had supplied EU vaccine stock to the UK, launched plans to force all EU vaccine manufacturers to ask for permission before exporting jabs out of the bloc.
On Jan 29, the President of the European Commission, Ursula von der Leyen, took the controversial decision to invoke Article 16 of the Northern Ireland Protocol to prevent the protocol from being used as a "back door" to bypass restrictions the EU had placed on vaccine exports from leaving the bloc.
The First Minister of Northern Ireland, Arlene Foster, described the move as an "incredible act of hostility", whilst the Boris Johnson expressed "grave concerns" over the EU's decision and called Irish Prime Minister, Micheal Martin. Mr Martin said that he had no prior knowledge that the EU intended to invoke Article 16 and initially heard about it through a public statement.
Just hours later, a U-turn came from the EU where they said that the decision to trigger Article 16 was an "oversight", after widespread condemnation from leaders over the move.
Mrs von der Leyen later tweeted that she had spoken to Mr Martin and had "constructive talks" with Mr Johnson.
Constructive talks with Prime Minister @BorisJohnson tonight.
We agreed on the principle that there should not be restrictions on the export of vaccines by companies where they are fulfilling contractual responsibilities.— Ursula von der Leyen (@vonderleyen) January 29, 2021
Although, Mrs von der Leyen admitted the move was an error, she has refused to take personal responsibility for the mistake.
AstraZeneca has now agreed to supply the EU with nine million additional vaccines by the end of March, however, it seems as if some of the issues are still on going.
On March 18, the EU announced that it was going to send a formal letter to AstraZeneca in an attempt to resolve its dispute over vaccine supplies after the company has said it will not be able to meet its contractual commitment of 300m doses for the EU in the first half of the year but instead will be able to supply only 100m due to production problems and export restrictions.
The UK and EU issued a joint-statement on Mar 24 insisting that they are working together "to create a win-win situation and expand vaccine supply for all our citizens".
How many doses does the UK have?
The UK has secured 100 million doses of the Oxford/AstraZeneca vaccine, this is the most the Government has ordered out of all of the potential vaccine candidates. The order is enough to vaccinate 50 million people.
AstraZeneca said it aimed to supply millions of doses in the first quarter of this year as part of an agreement with the Government. There will be four million doses available post-authorisation and tens of millions of doses in the first quarter of this year.
A specific schedule is difficult to establish as batches need to be quality approved by the MHRA.
Does it differ to Pfizer and Moderna's vaccines?
Yes. The jabs from Pfizer and Moderna are messenger RNA (mRNA) vaccines.
Conventional vaccines are produced using weakened forms of the virus, but mRNAs use only the virus's genetic code.
An mRNA vaccine is injected into the body where it enters cells and tells them to create antigens.
These antigens are recognised by the immune system and prepare it to fight coronavirus.
No virus is needed to create an mRNA vaccine. This means the rate at which the vaccine can be produced is accelerated.
The Pfizer and Moderna vaccines have both been approved in the US.
The UK Government began the roll-out of the Pfizer vaccine on Dec 8.
Unlike the Pfizer vaccine, the Oxford jab does not require ultra-low temperatures.
The Oxford jab requires temperatures between 2C and 8C and can be stored for at least six months.
This is the typical temperature of a domestic refrigerator and this will make deployment of the vaccine much easier and faster.
What about antibodies and T-cells?
The Pfizer, Oxford/AstraZeneca and Moderna vaccines have been shown to provoke both an antibody and T-cell response.
Antibodies are proteins that bind to the body's foreign invaders and tell the immune system it needs to take action.
T-cells are a type of white blood cell which hunt down infected cells in the body and destroy them.
Nearly all effective vaccines induce both responses.
The Oxford/AstraZeneca vaccine induces robust antibody and T-cell responses across people of all ages, the data indicates.
Where is it being manufactured?
While there are some doses coming from Europe in the very first instance, the majority will be provided from the UK supply chain.
In an exclusive report on Jan 16, The Telegraph shared plans for a new £158m “super-factory”, which would produce 70m doses of an emergency vaccine on British soil- enough to vaccinate the entire nation against new coronavirus strains within four months. The factory will open later this year.
Do you need two doses of the Oxford vaccine?
The MHRA has recommended the over-18s should receive two doses to be administered with an interval of between four and 12 weeks.
When will I get my second dose?
The Government announced on Dec 30 that it was delaying the second dose of every vaccine in order to reach as many people as possible in the first round of vaccinations.
Both the Oxford vaccine and the Pfizer/BioNTech jab will be given to people as one shot, followed by another up to 12 weeks later, in order to extend some protection to as many people as possible as quickly as possible.
This is not without controversy, however.
The government's Joint Committee on Vaccination and Immunisation (JCVI) says unpublished data suggests the Oxford-AstraZeneca vaccine is still effective with doses 12 weeks apart - but Pfizer has said it has tested its vaccine's efficacy only when the two doses were given up to 21 days apart.
The World Health Organisation has recommended a gap of four weeks between doses - to be extended only in exceptional circumstances to six weeks.
Will the vaccines be given out 24 hours a day?
In a further bid to accelerate vaccination, Boris Johnson has announced that 24-hour vaccine centres will be opened "as soon as we can”, with the the head of NHS England confirming on Jan 17 that several hospitals will trial 24/7 vaccine centres.
Matt Hancock, however, told BBC Breakfast a 24/7 approach was unlikely to be "the major factor" in hitting the mid-February target, but he was "absolutely" behind it "if it helps speed things up".
Sources in Whitehall have said that plans are in place to pilot a 24-hour vaccination centre to test demand. This comes as manufacturing companies have told ministers that they will not yet be able to produce enough vaccines should 24-hour roll out be introduced across the country.
Supplying vaccinations overnight will speed up the rollout, and allow the Government to reach their goal of vaccinating 32 million people- 60 per cent of the UK adult population by April 15.
Can pregnant women have the vaccine?
Pregnant women and breastfeeding mothers have now been given the green light to take either the Oxford and Pfizer coronavirus vaccines following an appropriate case-by-case risk evaluation with their healthcare practitioner.
This is a reversal of previous advice which was put in place as precautionary measure.
Traditionally pregnant women are not included in clinical trials, but following a review the MHRA are recommending pregnant women be given the opportunity to receive the vaccine as as there is no evidence they would be at risk.
Dr June Raine, chief executive of the MHRA, said: "Our advice to date has been that given that in initial lack of evidence on a precautionary basis, use of a vaccine wasn't recommended in pregnancy and women with breastfeeding should not be given the vaccine.
"But now that we have reviewed further data that has become available, the Commission on Human medicines has advised that the vaccine can be considered for use in pregnancy when the potential benefits outweigh the risks following an individual discussion with every woman."
Can people with allergies have the vaccine?
The rollout of the Pfizer vaccine was temporarily halted for those who are known to suffer from severe allergic reactions following a handful of adverse events in the initial distribution of the vaccine.
There were some concerns that this would also apply to the Oxford jab.
However, following a review, the UK regulatory body has recommended both the Pfizer and Oxford vaccine are safe to administer to those with food or medicine allergies.
Only those who have a known history of reacting to vaccines in the past should proceed with caution.
Sir Munir Pirmohamed, clinical pharmacologist and geneticist, and chairman of Commission on Human Medicine Expert Working Group, said. "We've come to the recommendation people with a known history of reacting to any specific ingredients of vaccine should not have it. But people with allergies to other medicines or food can have the vaccine."
Dr June Raine added that "at least 800,000 in the UK, probably a million and a half in the US" have already received the Pfizer vaccine.
There has been "no additional concerns and this gives us further assurance that the risk of anaphylaxis can be managed through standard clinical guidance and an observation period following vaccination of at least 15 minutes.
Read more: The priority list for the Oxford and Pfizer vaccines - and how they will be rolled out
Have you had the Covid vaccine or are you due to get one? We want to hear from you. Get in touch with us here.

