Oxford coronavirus vaccine results leave regulators with 'dilemma', warns scientist

The Oxford vaccine may not be as useful as first thought, after published results showed that high efficacy rates cannot be substantiated.
At the end of November, Oxford and AstraZeneca announced they had efficacy rates of 90 per cent when people were given a half dose, followed by a full dose several weeks later.
However when people received two full doses, efficacy fell to just 62 per cent, far lower than the mid-90s results shown by Pfizer and Moderna.
The results leave regulators with a "dilemma", scientists have warned. The Oxford data is currently being studied by the Medicines and Healthcare products Regulatory Agency (MHRA) which will decide whether to approve the jab.
Watch: Oxford Covid-19 vaccine has good safety record and efficacy – study
However, new data published in The Lancet on Tuesday has led to fears that the more effective half-dose regime will not be approved.
The report showed the half-dose trial only "provides a suggestion" that it would be more effective, and said more research is needed. The half-dose vaccine has also not yet been proven to work in the over-55s, so it may not be useful in the first wave of vaccinations.
And even if the two dose regime is approved, the study shows it only plays a small role in preventing transmission, which may make it unsuitable to be rolled out to the general population.
Experts said regulators would now be in a quandary about whether to approve the low dose vaccination, or allow it to be used in older people.
Dr Simon Clarke, associate professor in cellular microbiology, University of Reading, said: “The report on the successful trial of the Oxford/AstraZeneca presents regulators with something of a dilemma.
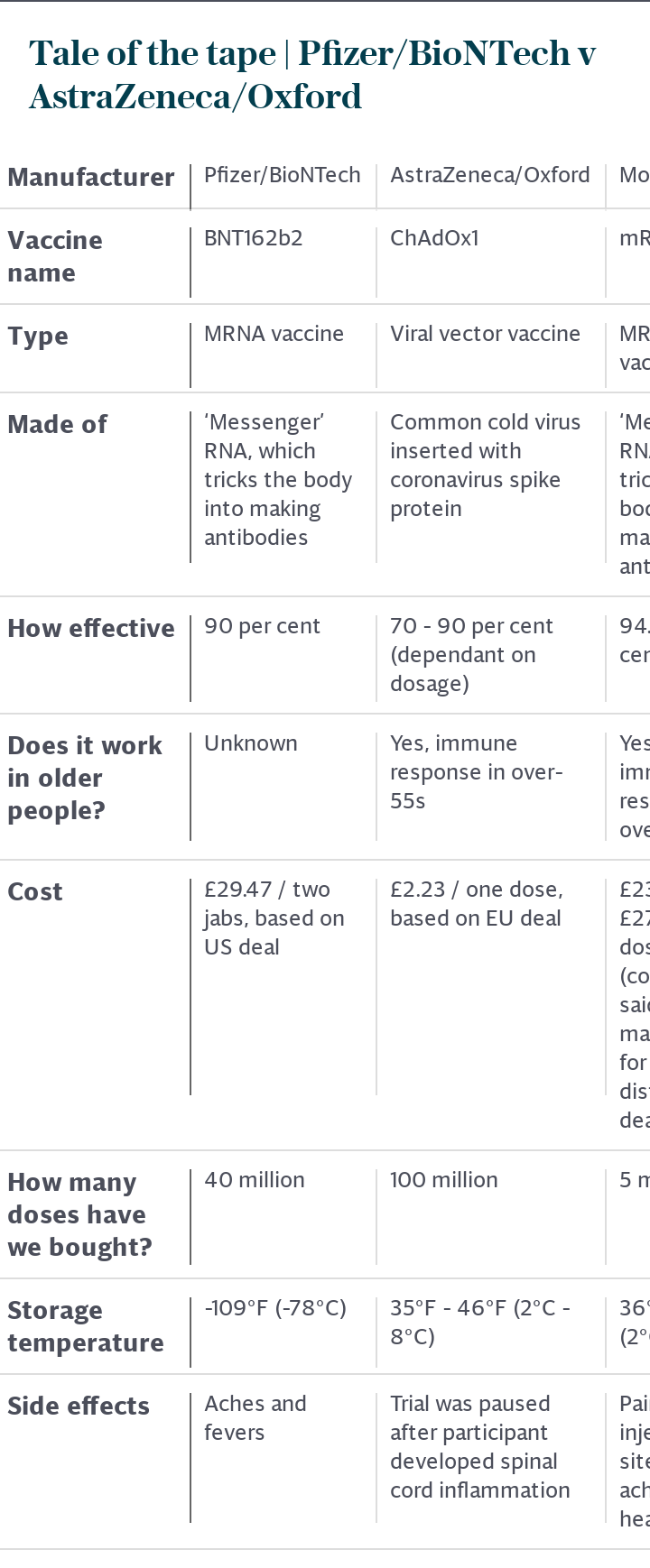
“Data are most compelling for the cohort who got half a dose of the vaccine in their first jab. Not only does this seem to confer greater protection against disease, it is in this group that there is a reduction in asymptomatic transmission of the virus, something which is essential if herd immunity is to be obtained to get wider protection of the population.
“Unfortunately, this cohort was relatively small, reducing the reliability of the findings – moreover it did not contain any older participants (age 55 or over) and it remains possible that if the regulators allowed the vaccine to be used in this manner, the most at risk group may not be protected.”
Dr Julian Tang, honorary, clinical virologist, Respiratory Sciences, University of Leicester, added: “A lower vaccine efficacy does run the risk of leaving a large proportion potentially unprotected despite being vaccinated.
“Those who have been vaccinated and think they are immune may behave more freely which may serve to spread the virus further if they do become infected.
“The other finding is that there is insufficient data as yet to confirm the vaccine efficacy against those over 55 years old. So if it is licensed by the MHRA, will it just be for the standard dose regimen but only for those aged 18-55 years? Or will they assume some level of efficacy (under 50 per cent) in those over 55 years also, given the current Covid-19 pandemic crisis?”
At a briefing ahead of publication in The Lancet, the vaccine team from Oxford University said they hoped to see regulatory approval "in the coming weeks".
But Prof Andrew Pollard, professor of paediatric infection and immunity, University of Oxford, appeared to suggest that it would not be enough on its own to bring the pandemic under control and that other vaccines would be needed.
“I think what we need are vaccines in people's arms and I personally think we need to be completely agnostic about which vaccine we use, because we want multiple vaccines, as soon as possible.
“This really can’t be a competition between developers, this has to be a competition against the virus.”
Watch: Quarter of Brits believe coronavirus conspiracies
Mene Pangalos of AstraZeneca also said he believed even a vaccine of 62 per cent efficacy would be useful in preventing severe Covid and death.
“I do believe that a vaccine of 62 per cent, which is the minimum we have shown, is still a viable and effective vaccine.
“We have no cases of severe Covid or death so in terms of the impact it will have on the healthcare system done on populations at large. I think it's very important.”
The Lancet study found that the Oxford vaccine "has a good safety record and efficacy" following trials on 11,636 volunteers across the UK and Brazil.
Prof Sarah Gilbert, professor of vaccinology at the University of Oxford, said: “We hope that this vaccine will shortly be in use to start saving lives.”

