Pfizer/BioNTech's COVID Jab Gets CHMP Nod for Kids Aged 5-11
Pfizer PFE and partner, BioNTech BNTX announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use (“CHMP”) has given a positive opinion recommending extended use of their COVID-19 vaccine, Comirnaty, in children five to under 12 years of age in Europe. Comirnaty is already approved for administration in adults and children aged 12 and above in Europe.
The CHMP’s recommendation will be reviewed by the European Commission, which should soon make its decision on expanding the conditional marketing authorization (CMA) to allow the vaccine’s use in this age group before vaccinations begin. If approved, Comirnaty will be the first vaccine to be approved for administration in this age group in Europe. The dose of Comirnaty for kids in this age group will be lower than that for people aged 12 and above (10 µg compared with 30 µg), and the vaccine will be given as two injections, three weeks apart.
Pfizer’s stock has risen 38.3% this year so far compared with an increase of 14.6% for the industry.
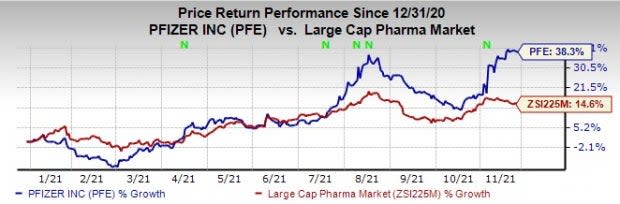
Image Source: Zacks Investment Research
Germany-based BioNTech’s shares are up 273.8% this year so far against the industry’s decrease of 17.3%.
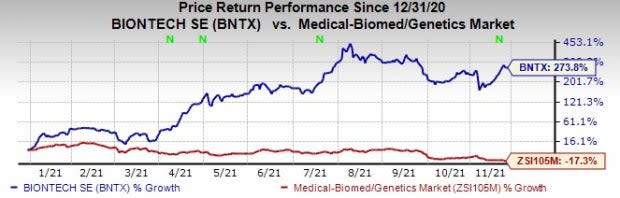
Image Source: Zacks Investment Research
Pfizer/BioNTech’s Comirnaty was approved for administration in children of this age group in the United States in October. The approval in the United States, as well as CHMP’s recommendation, was based on data from a phase II/III study, which showed that its COVID-19 vaccine had a favorable safety profile and generated robust immune responses and a vaccine efficacy rate of 90.7% in kids of this age group. Based on this data, the CHMP concluded that the vaccine’s benefits outweighed its risks in children aged five to 11.
With a new wave of infections sweeping across Europe, immunizing school-going kids has become extremely important to reduce the spread of infection. In some European countries, children account for most cases, and their timely vaccination could be the key to bringing this new wave of pandemic under control.
Pfizer/BioNTech’s vaccine is being studied in even younger children. Separate studies are ongoing for children two to
Among other vaccine makers, Moderna’s MRNA vaccine remains available for emergency/conditional use in several countries. Moderna’s vaccine is authorized for use in adolescents in Europe, Canada and many more countries. A similar authorization in the United States is expected in January 2022. For children in the six to 11 age group, Moderna has filed an application in Europe seeking approval to expand the CMA for its COVID-19 vaccine to allow its use in children of this age group.
J&J’s JNJ adenovirus-based single-shot COVID-19 vaccine candidate is approved on emergency/conditional basis for adults 18 and older in several countries, including the United States and Europe.
A booster shot of J&J’s vaccine was granted emergency use authorization (EUA) by the FDA last month for adults aged 18 and older and who have completed at least two months after the initial dose.
Both Pfizer and BioNTech have a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Johnson & Johnson (JNJ) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Moderna, Inc. (MRNA) : Free Stock Analysis Report
BioNTech SE Sponsored ADR (BNTX) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research
