Pfizer (PFE) to Supply 40M Doses of Coronavirus Vaccine to COVAX
Pfizer Inc. PFE, along with Germany’s BioNTech SE BNTX announced that they entered into an advance purchase agreement with COVAX for supplying up to 40 million doses of their COVID-19 vaccine, BNT162b2. The vaccine doses will be delivered throughout 2021 with first deliveries expected to begin in the first quarter, subject to the execution of supply agreements under the COVAX Facility structure.
Per the press release, COVAX is a global initiative coordinated by the Global Alliance for Vaccines and Immunization, the Coalition for Epidemic Preparedness Innovations and the World Health Organization that ensures easy and impartial access to COVID-19 vaccines for all countries, regardless of their income levels.
Moreover, Pfizer and BioNTech will provide the COVID-19 vaccine to COVAX at a not-for-profit price, since the COVAX Advanced Market Commitment will ensure that 92 low- and lower-middle-income countries get access to COVID-19 vaccines at the same time as higher-income countries do.
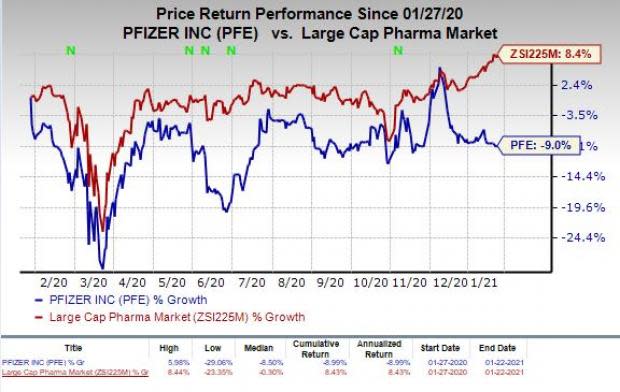
Shares of Pfizer have declined 9% in the past year against the industry’s increase of 8.4%.
In December 2020, the FDA granted emergency use authorization to Pfizer and BioNTech’s mRNA-based coronavirus vaccine candidate, BNT162b2. The vaccine is currently approved for emergency/temporary/conditional use in several countries, including the United States, 27 EU member countries, the United Kingdom and Canada. The emergency/temporary approvals for BNT162b2 came in less than a year since the companies started developing it, which is probably the fastest in the history of vaccines.
Several millions of doses of the vaccine candidate have already been administered in the United States. The subsequent payments for the vaccine doses are expected to boost Pfizer/BioNTech’s revenues/profits for the next few quarters.
Pfizer and BioNTech plan to file a biologics license application for possible full regulatory approval in 2021 as it gathers additional data from ongoing studies on BNT162b2.
Meanwhile, another mRNA-based COVID-19 vaccine candidate, mRNA-1273, developed by Moderna, Inc. MRNA, has been approved for emergency/temporary use in the United States, Canada, Europe, the United Kingdom, Israel and Switzerland.
AstraZeneca’s AZN adenovirus-based coronavirus vaccine candidate, AZD1222, developed in partnership with the University of Oxford, is also approved for emergency use in the United Kingdom, India and a few other countries.
Zacks Rank
Pfizer currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Just Released: Zacks’ 7 Best Stocks for Today
Experts extracted 7 stocks from the list of 220 Zacks Rank #1 Strong Buys that has beaten the market more than 2X over with a stunning average gain of +24.4% per year.
These 7 were selected because of their superior potential for immediate breakout.
See these time-sensitive tickers now >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Moderna, Inc. (MRNA) : Free Stock Analysis Report
BioNTech SE Sponsored ADR (BNTX) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research
