Pregnant women were kept out of clinical trials. That left them vulnerable to COVID-19
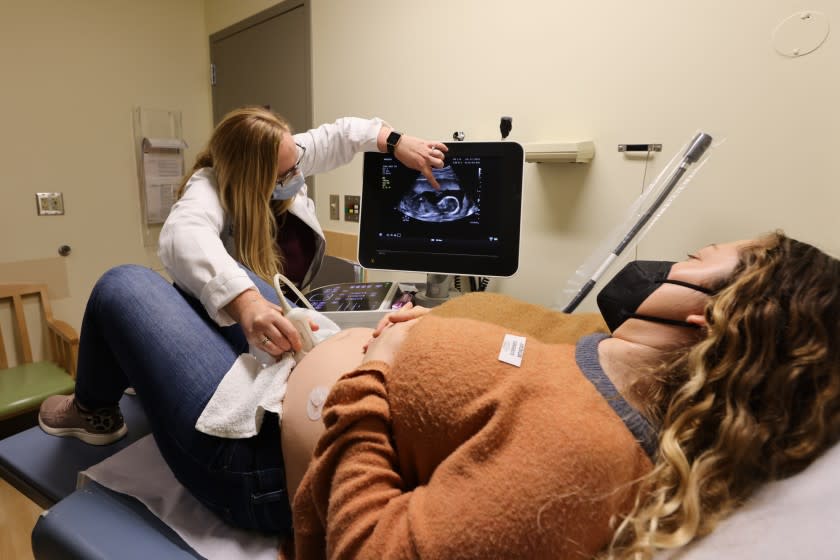
As the fast-spreading Delta variant filled the University of Washington Medical Center with COVID-19 patients this summer, Dr. Linda Eckert was struck by something: More pregnant patients were hospitalized with the disease than at any other time during the pandemic.
Expectant mothers were struggling to breathe. Some were on mechanical ventilators. A few didn't make it.
“I have rarely seen any condition confer this much risk to pregnant individuals,” said Eckert, an obstetrician-gynecologist with a specialty in infectious diseases. "It's actually just ... horrifying."
Experts say vaccination could have prevented most serious illnesses and deaths in the current surge. But that message was slow to get out to pregnant women due to a long-standing tradition of excluding them from clinical trials of experimental medicines — a practice that extended to COVID-19 vaccines.

As a result, for months after the vaccines became available, doctors and their pregnant patients had little relevant safety data to rely on. So they turned to each other in an effort to crowdsource their own best practices.
Some scoured regulatory filings, medical journals and websites for any information that might be relevant. Others joined registries of pregnant women who opted to get the shot so that researchers could track their health outcomes as well as those of their babies.
“It felt good to be a part of it,” said Dr. Emily Fay, a maternal-fetal medicine specialist in Seattle who enrolled in a registry while pregnant herself. “Hopefully it helps add to what we know.”
More than 22,000 pregnant people have been hospitalized with COVID-19 over the course of the pandemic, according to the Centers for Disease Control and Prevention, and as of early October, 171 had died. That includes 22 fatalities in August 2021, the highest toll of any month since the outbreak began.
Last month, the CDC issued a health advisory imploring women who are pregnant, trying to become pregnant or nursing to get vaccinated "as soon as possible."
Yet two-thirds of pregnant women remain unvaccinated, CDC data show.
::
The inclination to keep experimental drugs away from pregnant women is in many ways consistent with the spirit of the Hippocratic Oath, in which doctors pledge to "do no harm" to the patients under their care.
“You think that you're actually doing the right thing because you're concerned about the exposure to the baby,” said Dr. Laura Riley, a maternal-fetal medicine specialist at Weill Cornell Medicine.
But that way of thinking ignores the fact that in some cases, a baby — and its mother — could wind up being harmed if access to a much-needed medication is withheld, delayed or administered at the wrong dose.
“The prevailing idea is that pregnant people need to be protected from research,” said Dr. Diana Bianchi, head of the National Institutes of Health's Eunice Kennedy Shriver National Institute of Child Health and Human Development. “It's a very paternalistic attitude, and we are trying to change the culture, to protect pregnant people through research, instead of from research.”
Such a shift would be welcomed by the many doctors and researchers who’ve been laying the foundations for such a change.
Bianchi and her colleagues convened a task force of over two dozen experts from a variety of fields that worked for more than two years on recommendations for conducting research in pregnant and lactating women.
Riley, who was not on the task force, agreed that pregnant women could be included in studies in a staged manner without subjecting them to undue risk. For example, they could start with tests in pregnant animals, then move to women in their third trimester, when the fetus is in its final stages of growth and development. If all goes well, they can work backward to women in earlier stages of pregnancy.
“I've always found it really annoying to listen to people say, ‘Well, we couldn't possibly test that in pregnancy,’” she said.
The task force's advice has been public since September 2018 — and to ensure that it “didn't just sit on a shelf,” the group met several times to come up with advice on how to implement it, Bianchi said.
Yet even with a road map available, pregnant people were still barred from both Pfizer's and Moderna's first clinical trials for their COVID-19 vaccines.
Both companies gave pregnancy tests to potential trial participants and dropped anyone who tested positive. A small number of women — 23 in the Pfizer trial and 13 in Moderna's — had pregnancies that were missed by the screening tests or that began after they got their injections, but they were too few to produce significant results.
“When it came to the rubber meeting the road with pregnant women and vaccines," Bianchi said, "it appeared that no one had really paid attention to our recommendations.”
Pfizer representative Kit Longley said the company followed guidance provided by the Food and Drug Administration, "including the important consideration [of] whether and when to enroll pregnant [women] and women of childbearing potential." The Pfizer vaccine is currently being tested in pregnant women.
Moderna, which is conducting an observational study of vaccinated pregnant women, did not respond to a request for comment.
::
Fay, who works at the University of Washington Medical Center, said she'd had no doubt she’d be getting the COVID-19 vaccine when she became eligible for it in late December. It was during the winter surge, and hospital beds were filling up.
She'd followed the preliminary research on the vaccine closely. She knew that her life, and the lives of her most vulnerable patients, were at stake.
Still, around 17 weeks into her pregnancy, she couldn’t help but feel a prick of nerves.
“I felt like this was the right thing to do, but there's just always a fear of the unknown,” Fay said.
What kept her resolute was that in her own practice, she had witnessed firsthand how dangerous COVID-19 is for pregnant people.
Many changes during pregnancy may account for the higher risk. Among them: COVID-19 causes severe lung disease even as the growing uterus limits lung capacity by pushing against the diaphragm. Pregnancy also puts more strain on the cardiovascular system, which must pump a higher volume of blood around the body.
A study in JAMA Network Open of nearly 870,000 women who gave birth during the pandemic’s first year found that those with COVID-19 were nearly six times as likely to be admitted to an intensive care unit, more than 14 times as likely to require respiratory intubation and mechanical ventilation, and more than 15 times as likely to die as new mothers who did not have COVID-19.
Early in the vaccination campaign, the CDC said that pregnant people “may choose to” get the vaccine, framing it as a personal decision.
The World Health Organization initially went in the opposite direction. The WHO said it did not recommend COVID-19 vaccines for pregnant people unless they were at high risk of coronavirus exposure — a move that alarmed both the American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine.
“ACOG and SMFM continue to stress that both COVID-19 vaccines currently authorized by the U.S. Food and Drug Administration should not be withheld from pregnant individuals who choose to receive the vaccine,” the organizations said in a joint statement at the time.
Both societies would go on to recommend in July that pregnant people get vaccinated. And after releasing positive safety data in August, the CDC strongly encouraged them to get the shots.
The lack of clear data and strong recommendations earlier in the process may have left women who are pregnant or trying to become pregnant more vulnerable to another danger: vaccine misinformation.
It's an issue compounded by pregnant women's tendency to be cautious about what they put in their bodies, from what foods they eat to what medications they take.

“I think that is in general a good reflex,” Eckert said. “But I think in this instance it has made it very difficult to swing the pendulum.”
That inertia has had tragic consequences, with reports emerging from around the country of women who put off the vaccine because they were pregnant or trying to conceive and ended up severely ill or even dying — sometimes soon after giving birth.
::
Sara Nizzero was expecting her first child when the FDA issued its first emergency-use authorization for the Pfizer-BioNTech vaccine. The Houston-based medical researcher pored over the data for more than two months, ultimately concluding the vaccines were safe.
She wondered: How could busy pregnant moms without similar expertise figure this out themselves?
So in January, Nizzero started an “evidence-based” vaccine forum on Facebook for people who were pregnant, breastfeeding or trying to conceive. She shares the latest research and explains how the vaccines work. She and a group of moderators also screened posts to ensure they had solid scientific sources and did not spread misinformation.
Women have signed up in droves. The forum has amassed well over 80,000 members, and it continues to grow.
“With all the anti-vax propaganda that’s been coming up in the last years … there's a craving to actually access reliable knowledge,” she said.
That information — along with the photos of group members posting anatomy scans or photos of newborns after getting the vaccine — helped seal the deal for Maggie Snyder, a communications professional in Minnesota.
“It’s scary when you’re not making the decision for just you,” said Snyder, who got her first dose of Pfizer when she was 15 weeks into her pregnancy.

Dr. Alisa Kachikis opted for a more forward-looking type of research. The maternal-fetal medicine specialist started a registry of pregnant people who’d gotten the vaccine so she could track their health outcomes as well as those of their babies. Fay, her officemate, quickly signed up.
Fay wasn’t the only one. The registry, originally a local effort, drew so much interest that it began enrolling women from all over the U.S. and beyond. Tens of thousands joined, and their collective experience has helped demonstrate the safety of the shots in results published in August in JAMA Network Open.
Indeed, many of the findings on COVID-19 vaccines and pregnancy are thanks to women who took the plunge before authorities decisively weighed in. Among them: a study of 36 infants in the American Journal of Obstetrics & Gynecology Maternal–Fetal Medicine that found all of the newborns whose mothers were vaccinated during pregnancy had protective antibodies at birth.
"It's wonderful that so many pregnant people are participating in the research," Bianchi said. "It's not for lack of interest that pregnant people aren't included."
Bianchi is cautiously optimistic that pregnant people will be more effectively included in research the next time a major disease outbreak hits the United States.
"I'm hoping that's a lesson that was learned during this pandemic," she said.
This story originally appeared in Los Angeles Times.

