All the States of Matter You Didn't Know Existed




All the States of Matter You Didn't Know Existed
Before scientists discovered the new state of matter last week, we were basically all used to just three states of matter. After all, during our daily lives we encounter some variety of solids, liquids and gases. Solids hold a definite shape without a container, liquids conform to the shape of their container, and gases not only conform to a container, but also expand to fill it.
And there's variety amidst these three: A crystalline solid, for example, has all its atoms lined up in exactly the precise order in perfect symmetry, while a quasicrystal solid fills all its space without the tightly regulated structure. Liquid crystals, which make up the visual components of most electronic displays, have elements of both liquids and crystal structures, as anyone who has ever pushed the screen of their calculator can confirm.
Under standard conditions on Earth, solids, liquids and gasses are the vast majority of what a person will experience in life. But that doesn't mean there's not a whole lot more beneath the surface.
In 1856, the legendary British scientist Michael Faraday was studying thin sheets of gold leaf. Studying the properties of light and matter, Faraday was determined to make gold thin enough that it would be transparent to light. There wasn't machinery in existence at the time that could make films of gold that small, so Faraday had to use chemicals.
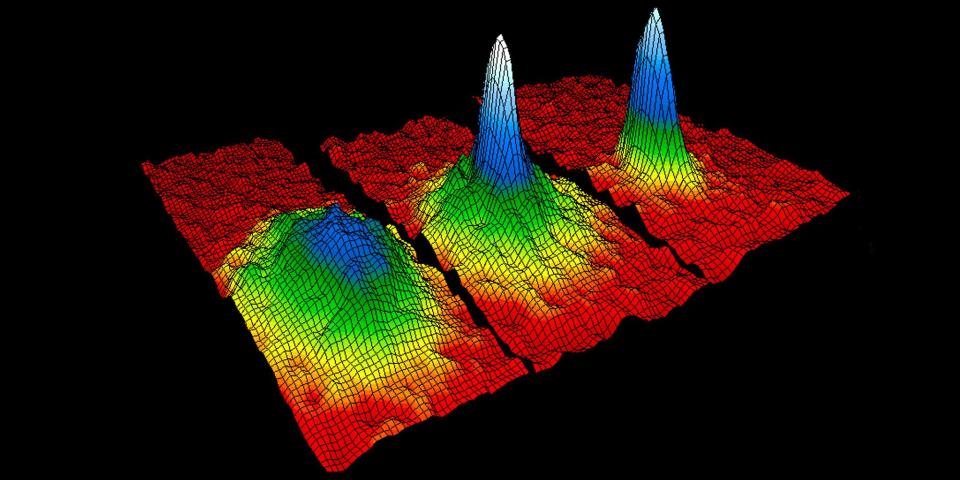
While washing the gold in these chemicals, Faraday noticed that the action produced a faint ruby colored fluid. This mixture of chemicals and gold, called a colloid, had the ability to scatter light shone in its direction. Even though scientific instruments at the time couldn't prove it, Faraday knew this light scattering was because of gold particles within the fluid. This was likely the first ever documentation of how a quantum state could have altering properties.
Quantum mechanics, quantum physics, and quantum computing study a wide variety of things, but they're all focused on what isn't noticeable to the naked eye. Faraday's light-scattering gold particles were in a quantum state—the smallest they could possibly exist. Since the 1850s, the science and exploration of quantum states has, with a little bit of irony, grown tremendously, and has revealed things previously thought unimaginable. Take, for example, topological superconductivity, that aforementioned new state of matter.
Through quantum studies, space exploration, and several other fields, there have been several discoveries of new types of matter, such as the five that follow. Not all of them are useful, although some are quite practical. But they've all shown that existence is more than three states.
Solids, liquids, and gases are just the beginning.
